  
Lower right: In the Auger effect, the energy absorbed when the outer electron replaces the inner electron is transferred to an outer electron. 
An x-ray, equal in energy to the difference between the two electron shells, is emitted. Lower left: An outer electron replaces the "missing" electron. May undergo electron capture but not decay.Scheme of two types of electron capture. Neutrino is unique in that it interacts with matter only via the weak nuclearįorce, which is why it is so hard to detect. Weaker than the strong nuclear force that holds the nucleus together. Referred to as the weak interaction because it is 10 12 times Nuclear force that plays the crucial role. Postulated the existence of the fourth force in nature: the weak nuclear force. That could only have been initiated by the neutrino. Successfully identified the neutrino by detecting the products of a reaction The neutrino was not experimentally observed until 1956. In 1934, Italian physicist Enrico Fermi (1901 - 1954) named Pauli’s particle the neutrino ( ), meaning “ little neutral one ” in Italian, and formulated a theory of decay using this particle.įermi’s theory successfully explained all experimental observations.įor instance, the shape of the energy curve shown in figure (1) for Bi-210Ĭan be predicted from the Fermi Theory of beta decay. Hence, it would beĮxpected that the beta particles emitted would have a range of energiesĭepending on the energies of the neutral particles emitted with them. Particle and the neutral particle emitted with it. For each beta emission, the total energyĬarried away from the decaying nucleus would be shared between the beta This particle would have noĬharge and zero rest mass (hence, travel at the speed of light) but would Proposed that a neutral particle was emitted along with the particle. Laws of conservation of energy, linear and angular momentum, Austrian Careful measurements indicated that linear and angularīismuth-210 into polonium-210 where a neutron changes into a proton.ĭistribution of electrons emitted in beta decay (figure 1) and to satisfy the It was if the law of conservation ofĮnergy was violated. But it was foundĮxperimentally that the kinetic energy of the emitted electron could have any Hence, it was expected that the emitted electrons in beta decay would have aįixed kinetic energy in each beta decay transmutation. Small energy whereas the electron would gain most of the energy liberated. Much larger than that of an electron it would recoil only very slowly with Only the daughter nucleus and an electron. It was first envisaged that the products of beta decay were the Is produced and hence in the process energy is liberated as kinetic energy of When a nucleus captures (absorbs) one of its orbiting electrons Transmutation, an electron neutrino is also emitted.īesides and emissions, there is a third process The positron is identical to anĮlectron except that its charge is positive and is the antiparticle of the The particle called the electron antineutrino (antiparticle of the electronĪ free neutron can also decay but not a free proton (on averageĪ free neutron at rest lives for ~15 minutes) Indistinguishable from an orbital electron. We use theįor the electron to show its origin, nonetheless it is The electron is created in the nucleus itself. Is conserved a beta particle is emitted from the nucleusĪnd momentum are conserved a particle called a neutrino / antineutrinoĪlpha rays and move at a very high speed. Stability can beĪchieved by the conversion inside the nucleus of a proton into a neutron or a Relative to the number of protons present for stability. Now that a nucleus exists which has either too many or too few neutrons It combines with a proton, forming a neutron and a neutrino. During electronĬapture, an electron in an atom's inner shell is drawn into the nucleus where Process that unstable atoms can use to become more stable. Proton in an atom's nucleus turns into a neutron, a positron and an Neutron in an atom's nucleus turns into a proton, an electron and 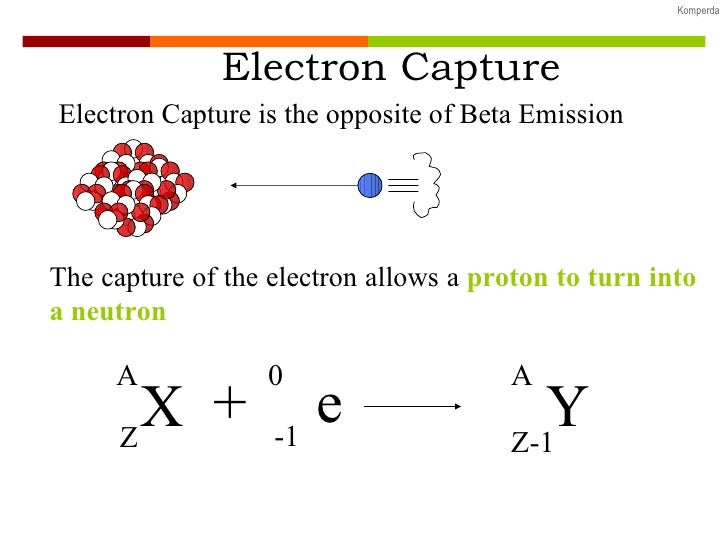
Unstable atoms can use to become more stable. Energy = rest energy + kinetic energy + potential energy 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
